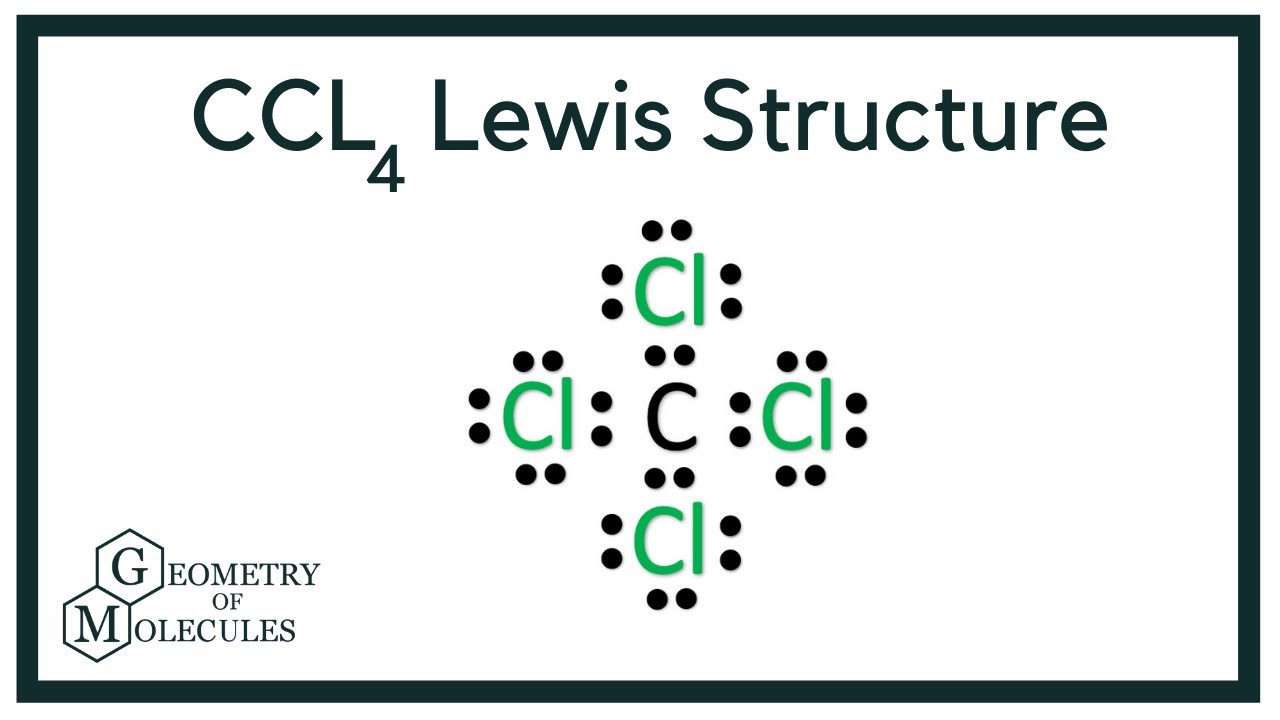
Draw The Lewis Structure For Ccl4
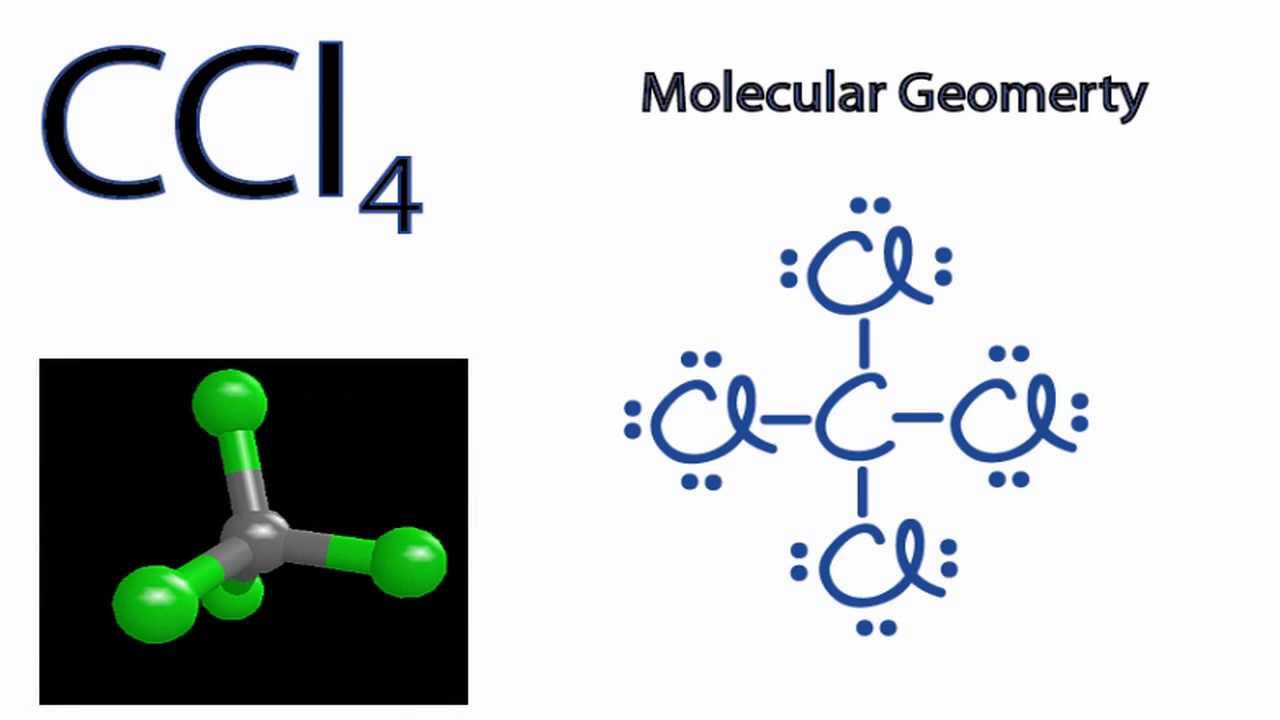
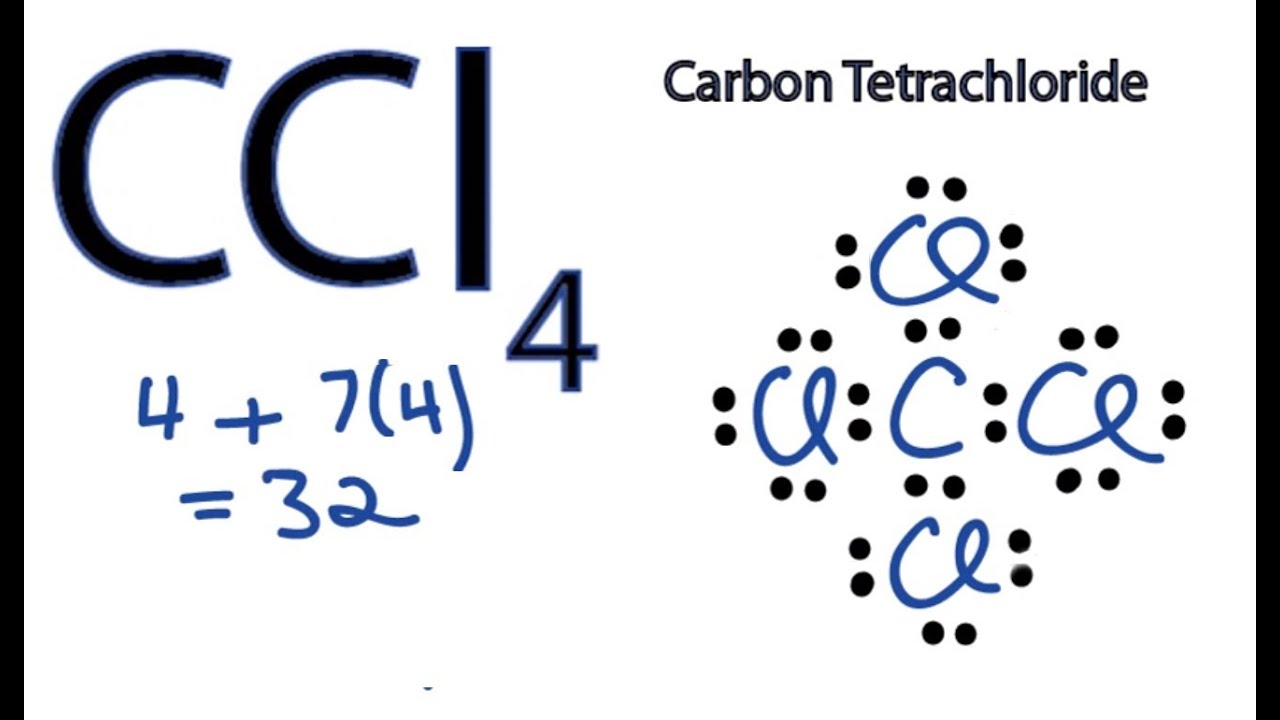
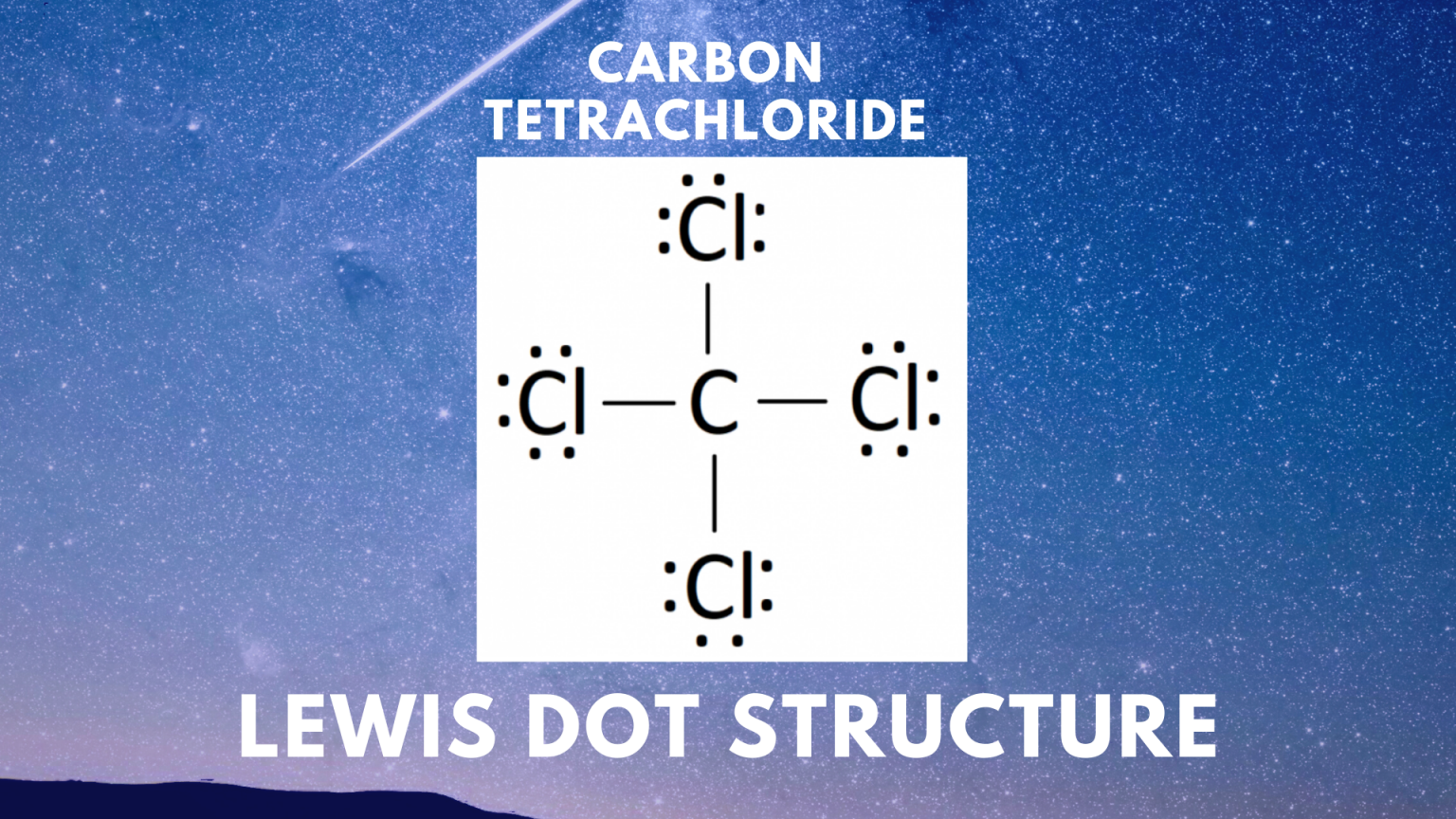
Draw The Lewis Structure For Ccl4 - Web for the lewis structure of ccl4 first, let’s calculate the total valence electrons. Draw lewis structures depicting the bonding in simple molecules Here are the steps that i follow when drawing a lewis structure. Determine total valence electrons to begin, we need to determine the total number of valence electrons […] ccl4 lewis structure. The lewis structure for ccl4 is a commonly tested lewis struc. In this tutorial, we’ll illustrate the steps to. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate. I also go over hybridization, shape and bond angle. Web to draw the lewis structure for ccl4, follow these steps: Learn how to draw the lewis structure of ccl4 with 32 valence electrons and 6 lone pairs. Web in this tutorial, we’ll illustrate the steps to draw the lewis structure for tetrachloromethane, denoted as ccl4. Draw lewis structures depicting the bonding in simple molecules Decide which atom is the central atom in the structure. Calculate the total number of valence electrons in ccl4: Carbon is in group 4 or 14, so it has. Web let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet. I also go over hybridization, shape and bond angle. We'll start by looking at the valence electrons. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Lewis structures are representations of molecules that include not only what atoms are present. Web drawing lewis structures for molecules with one central atom: Decide which atom is the central atom in the structure. Web draw lewis structures depicting the bonding in simple molecules. That will be the least. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Carbon (c) has 4 valence electrons, and. Each step of drawing is explained in detail in this tutorial. Web to draw the lewis structure for ccl4, follow these steps: The following procedure will give you the correct lewis structure for any. Web drawing lewis structures for molecules with one central atom: Web write lewis symbols for neutral atoms and ions; Web there are several steps to complete the lewis structure of ccl 4. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate. Draw lewis structures for covalent compounds. Decide which atom is the central atom in the structure. Web draw lewis structures depicting the bonding in simple molecules. Find total number of electrons of the valance shells of. The lewis structure for ccl4 is a commonly tested lewis struc. Web how to draw lewis dot structure of ccl4in this video you will learn how to draw lewis structure of carbon tetra chloride ccl4first find out valence electrons. If. That will be the least. Here are the steps that i follow when drawing a lewis structure. Web how to draw a lewis structure for ccl4? Find total number of electrons of the valance shells of. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Lewis structures are representations of molecules that include not only what atoms are present. Web drawing lewis structures for molecules with one central atom: Find total number of electrons of the valance shells of. Web learn how to draw the lewis dot diagram for ccl4 and understand its molecular structure and bonding with our clear and concise guide. Draw lewis. Each step of drawing is explained in detail in this tutorial. Calculate the total number of valence electrons in ccl4: Web drawing lewis structures for molecules with one central atom: Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Learn how to draw the lewis structure of ccl4 with 32 valence electrons and. Lewis structures are representations of molecules that include not only what atoms are present. Web in this tutorial, we’ll illustrate the steps to draw the lewis structure for tetrachloromethane, denoted as ccl4. Web how to draw a lewis structure for ccl4? Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Web for the. Web write lewis symbols for neutral atoms and ions; The lewis structure for ccl4 is a commonly tested lewis struc. Calculate the total number of valence electrons in ccl4: Web let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet. Web drawing lewis structures for molecules with one central atom: That will be the least. Web draw lewis structures depicting the bonding in simple molecules. Web to draw the lewis structure for ccl4, follow these steps: The following procedure will give you the correct lewis structure for any. Web let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate. Find total number of electrons of the valance shells of. The lewis structure for ccl4 is a commonly tested lewis struc. Web this video is intended to help students understand the lewis dot structure for carbon tetrachloride. Web let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet. Each step of drawing is explained in detail in this tutorial. Decide which atom is the central atom in the structure. Carbon is in group 4 or 14, so it has. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Lewis structures are representations of molecules that include not only what atoms are present. The following procedure will give you the correct lewis structure for any. I also go over hybridization, shape and bond angle. Web for the lewis structure of ccl4 first, let’s calculate the total valence electrons. Find out the hybridization, molecular geometry, and polarity of ccl4 with exa… Calculate the total number of valence electrons in ccl4: Web to draw the lewis structure for ccl4, follow these steps:CCl4 Molecular Geometry / Shape and Bond Angles YouTube
CCl4 Lewis Structure How to Draw the Dot Structure for CCl4 (Carbon
CCl4 Lewis Structure ,Valence Electrons ,Formal Charge ,Polar or Nonpolar
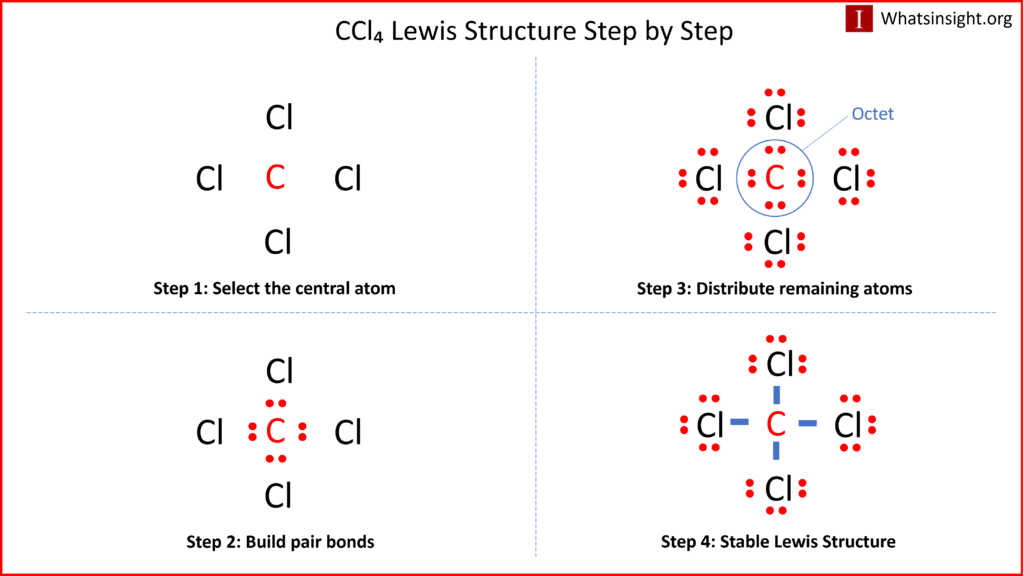
CCl4 Lewis structure in four simple steps What's Insight
Lewis dot structure of carbon Tetrachloride (CCl4). Scientific vector
CCl4 Lewis Structure Science Trends
How to Draw the Lewis Dot Diagram for CCl4 StepbyStep Guide
CCl4 Lewis Structure (Carbon Tetrachloride) YouTube
CCl4 Lewis Structure Science Trends
How to Draw the Lewis Dot Diagram for CCl4 StepbyStep Guide
Web Draw Lewis Structures Depicting The Bonding In Simple Molecules.
Carbon (C) Has 4 Valence Electrons, And.
Carbon Has Four Valence Electrons And Each Chlorine Atom Has Seven.
Web I Quickly Take You Through How To Draw The Lewis Structure Of Ccl4 (Carbon Tetrachloride).
Related Post: