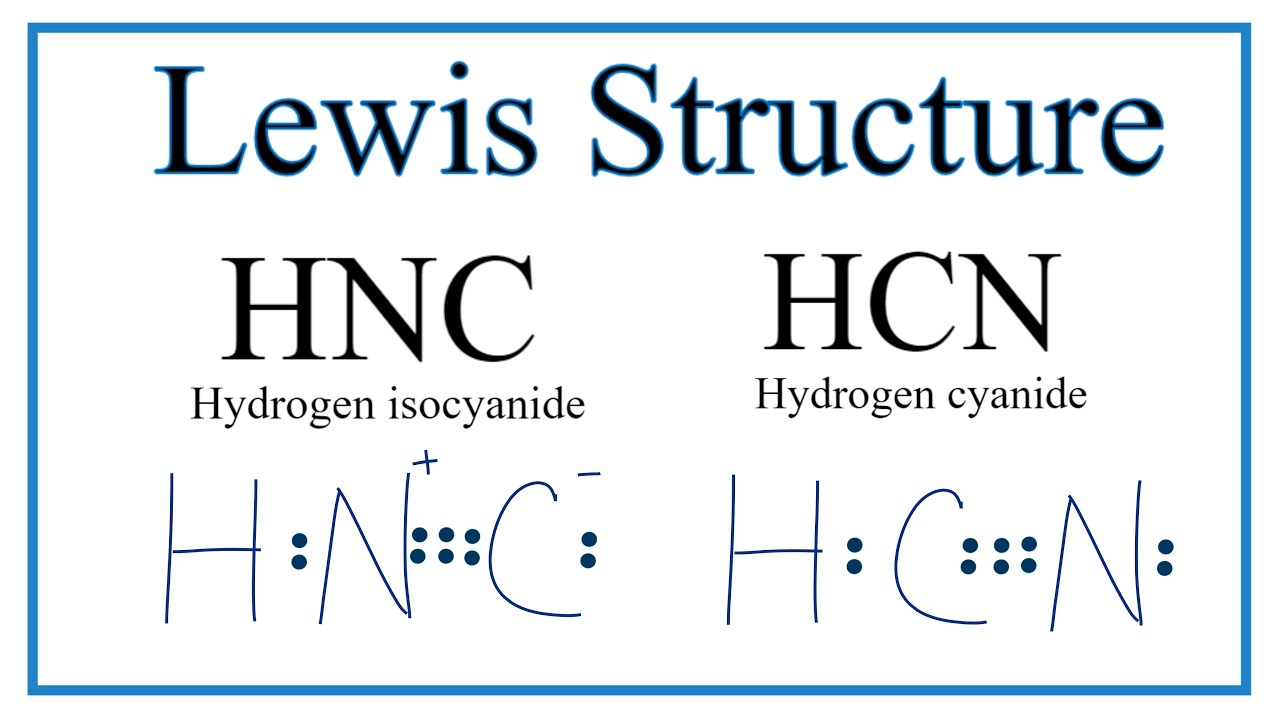
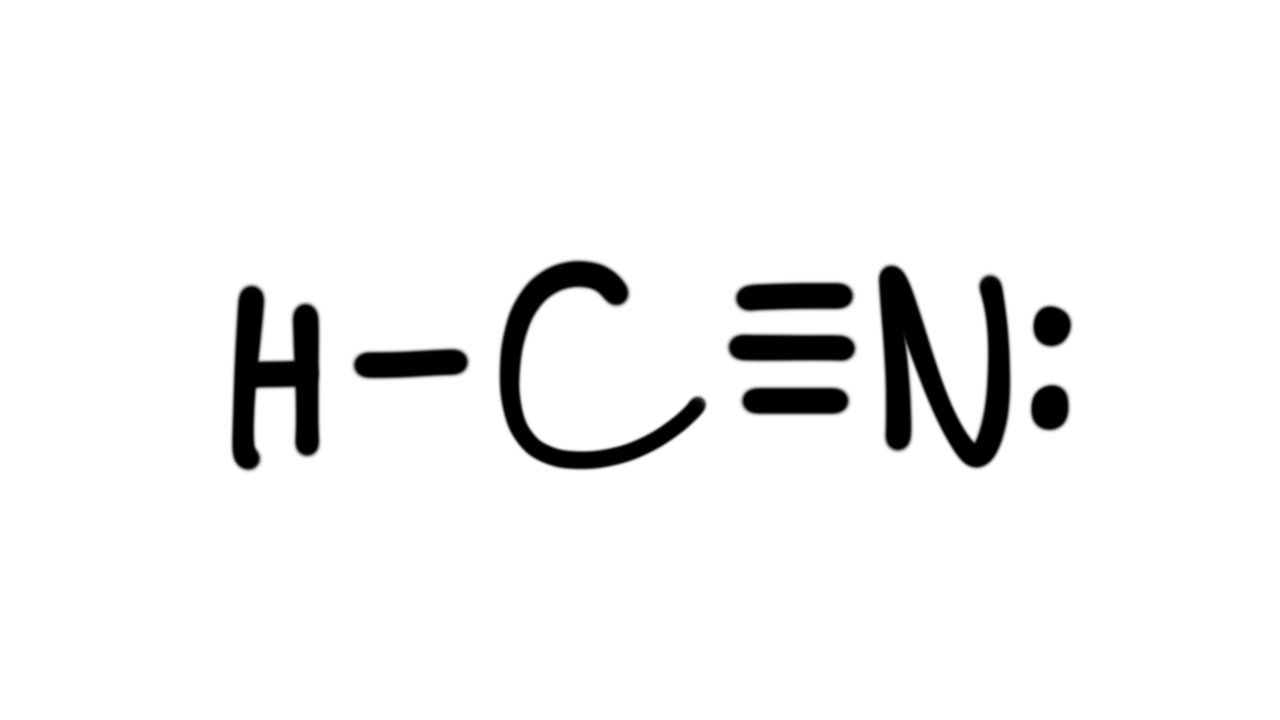Draw The Lewis Structure Of Hcn
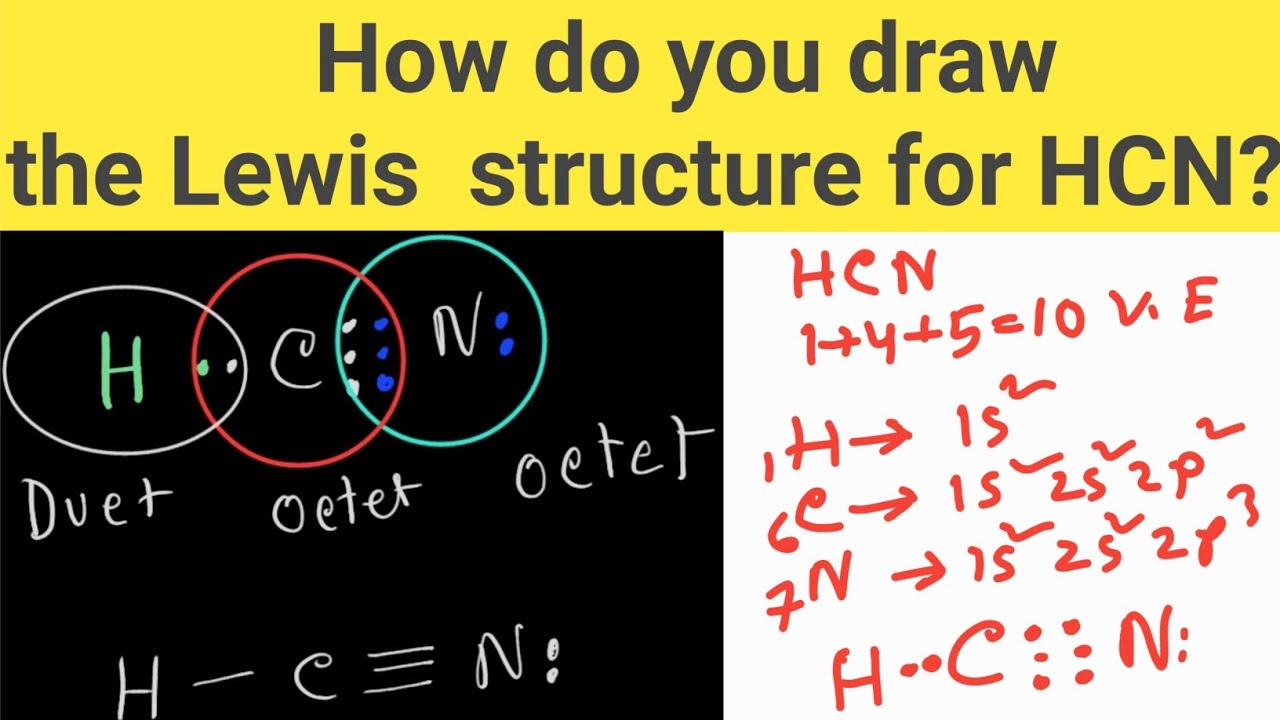
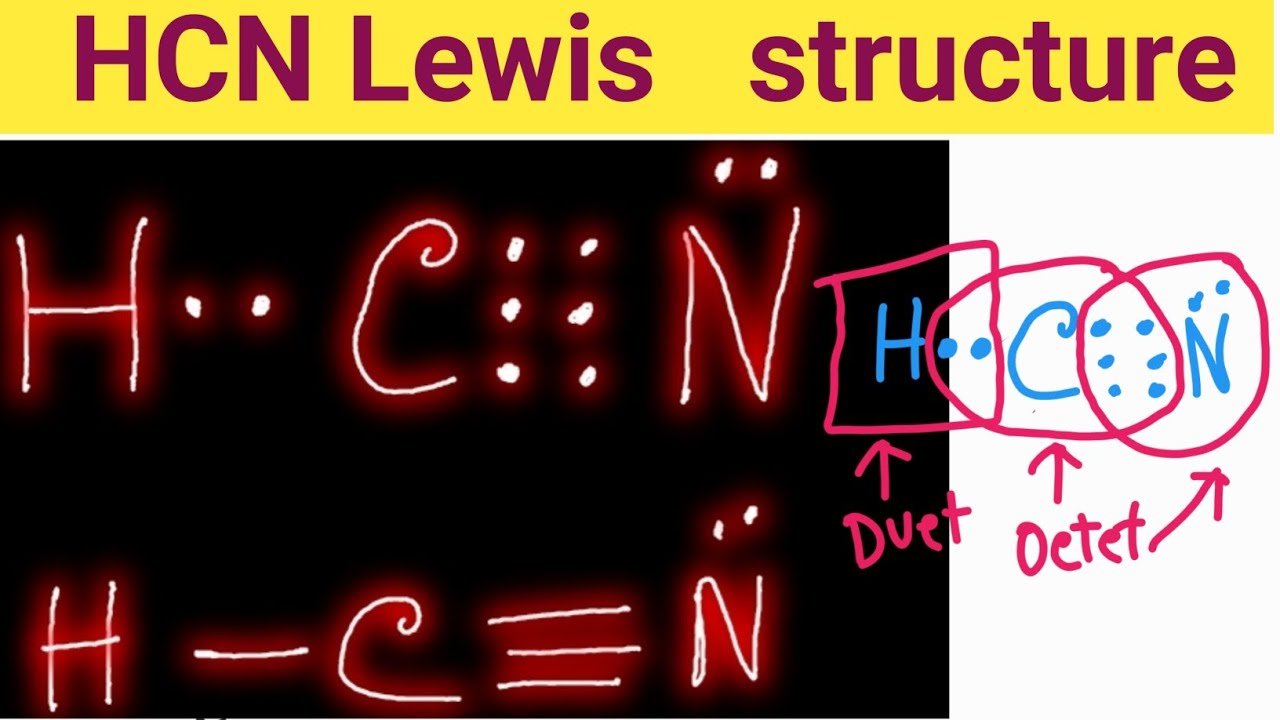
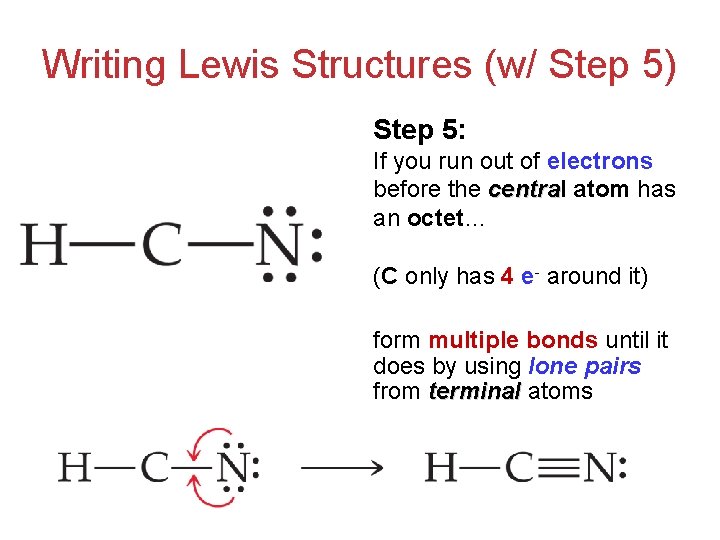
Draw The Lewis Structure Of Hcn - Web use these steps to correctly draw the hcn lewis structure: Web chemistry learning made easy.this tutorial will help you deal with the lewis structure and moleculargeometry for hydrogen cyanide (hcn). Some compounds have a very unique and different lewis structure and hcn is one of those. To begin, we can place a single bond between each. Calculate the total number of valence electrons. Web drawing the lewis structure for hcn. Lewis structures can also be useful in predicting molecular geometry in. For the hcn lewis structure, calculate the total number of valence electrons for the hcn. There is also a video and a study guide to help with other lewis dot problems. The hydrogen atom is bonded to the carbon atom, and the nitrogen atom is bonded to the carbon atom via a triple bond. Web hcn lewis structure: There is also a video and a study guide to help with other lewis dot problems. Here's how to do it. Web in this example problem, we draw the lewis structure for hcn, which has a triple bond. Put the least electronegative atom c in the middle with h and cl on either side. Web establish a general procedure for drawing lewis structures; For hcn, hydrogen has one valence electron, carbon has four valence electrons, and nitrogen has five valence electrons, making a total of ten valence electrons. Calculate the total number of valence electrons. Add the valence electrons of all the molecules' atoms: The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Lewis structures can also be useful in predicting molecular geometry in. How to draw the lewis structure for hcn. Web drawing lewis structures for molecules with one central atom: The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Calculate the total number of valence electrons. Draw lewis structures depicting the bonding in simple molecules Web use these steps to correctly draw the hcn lewis structure: = 4 + 1x4 = 8 valence electrons; Here, the given molecule is hcn. Web the nitrogen nucleus has 3 electrons from the triple bond, and 2 electrons from its lone pair, and 2 inner core electrons; Some compounds have a very unique and different lewis structure and hcn is one of those. Here's how to do it. The hydrogen atom is bonded to the carbon atom, and the nitrogen atom is bonded to the carbon atom via a triple bond. Web the nitrogen nucleus has 3 electrons from the triple bond, and 2 electrons from its. Put least electronegative atom in centre3. Web establish a general procedure for drawing lewis structures; Add the valence electrons of all the molecules' atoms: Hcn is a linear molecule, since it has only two electron groups and there are no lone pairs on the central carbon atom. Lewis structures can also be useful in predicting molecular geometry in. Web establish a general procedure for drawing lewis structures; There is also a video and a study guide to help with other lewis dot problems. Pcl 3 has 5 valence electros in p and 7 in each of the three cl: Web we show two methods to find correct lewis structure of hcn. Web to draw the hcn lewis structure,. To begin, we can place a single bond between each. Put least electronegative atom in centre3. For hcn, hydrogen has one valence electron, carbon has four valence electrons, and nitrogen has five valence electrons, making a total of ten valence electrons. Web in this video, we will look at the lewis structure of hydrogen cyanide having a chemical formula of. Web we show two methods to find correct lewis structure of hcn. = 4 + 1x4 = 8 valence electrons; Calculate the total number of valence electrons. Add the valence electrons of all the molecules' atoms: Carbon has 4 valence electrons, while each oxygen atom has 6. #1 first draw a rough sketch #2 mark lone pairs on the atoms #3 calculate and mark formal charges on the atoms, if required #4 convert lone pairs of the atoms, and minimize formal charges #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure Put the least electronegative atom c in the. Carbon has 4 valence electrons, while each oxygen atom has 6. Web the lewis structure of hcn shows that the carbon atom is the central atom and is covalently bonded to both hydrogen and nitrogen atoms. The hydrogen atom is bonded to the carbon atom, and the nitrogen atom is bonded to the carbon atom via a triple bond. Hcn. H + c + n =1 + 4 + 5 = 10. Web in this example problem, we draw the lewis structure for hcn, which has a triple bond. Web to draw the hcn lewis structure, follow these steps: Determine the total number of valence electrons by adding the valence electrons of each atom in the molecule. Some compounds have. Draw lewis structures depicting the bonding in simple molecules Hcn is a linear molecule, since it has only two electron groups and there are no lone pairs on the central carbon atom. Web in this video, we will look at the lewis structure of hydrogen cyanide having a chemical formula of hcn. Web use these steps to correctly draw the hcn lewis structure: How to draw the lewis structure for hcn. For the hcn lewis structure, calculate the total number of valence electrons for the hcn. Web chemistry learning made easy.this tutorial will help you deal with the lewis structure and moleculargeometry for hydrogen cyanide (hcn). #1 first draw a rough sketch #2 mark lone pairs on the atoms #3 calculate and mark formal charges on the atoms, if required #4 convert lone pairs of the atoms, and minimize formal charges #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure Web we show two methods to find correct lewis structure of hcn. In order to draw the lewis structure of hcn, first of all you have to find the total number of valence electrons present in the hcn molecule. Web 6 steps to draw the lewis structure of hcn step #1: Put the least electronegative atom c in the middle with h and cl on either side. The molecule is made up of one hydrogen atom, one carbon atom and a nitrogen atom. The carbon atom has (or shares) 3 electrons from the triple bond, and a lone pair of electrons, which it owns. You nave to put a triple bond between c and n. Pcl 3 has 5 valence electros in p and 7 in each of the three cl:Hcn Lewis Structure Bonds Draw Easy
Draw the Lewis dot structure of Hydrogen cyanide (HCN) molecule
HCN Lewis Structure, Molecular Geometry, Hybridization, MO Diagram, and
Estrutura De Lewis Hcn
Estrutura De Lewis Hcn ENSINO
Lewis Structure Of Hcn
Draw The Lewis Dot Structure Of Hydrogen Cyanide Hcn vrogue.co
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw
Hcn Lewis Structure Bonds Draw Easy
Lewis structure of HCN (Hydrogen cyanide) YouTube
Web Establish A General Procedure For Drawing Lewis Structures;
H + C + N =1 + 4 + 5 = 10.
To Begin, We Can Place A Single Bond Between Each.
There Is Also A Video And A Study Guide To Help With Other Lewis Dot Problems.
Related Post: